
Cooper DMF, Mons N, Karpen JW: Adenylyl cyclases and the interaction between calcium and cAMP signalling.Chen Y, Cann MJ, Litvin TN, etc: Soluble adenylyl cyclase as an evolutionarily-conserved bicarbonate sensor.This drug is widely used in studies aimed at dissecting intracellular signalling pathways. It thus appears that there is tight integration between cAMP and calcium, the cell's two major internal signallers.Īll known adenylyl cyclases are stimulated by exposure of cells to the diterpene forskolin. Also, ultrastructural labelling has demonstrated a close spatial association of adenylyl cyclases with sites of calcium entry in cells. Three of these are stimulated by calcium and two are inhibited. Of potentially great significance, five of the adenylyl cyclases known are regulated by calcium. More recently, it has become clear that cyclase activity is regulated by multiple effectors, which include not the alpha subunits of G s and G i proteins, but also the beta-gamma subunits of G proteins and protein kinase C. Also, when epinephrine binds to alpha-2 adrenergic receptors, adenylyl cyclase activity is inhibited, because that receptor is coupled to via G i, an inhibitory G protein. For example, the beta-adrenergic receptor is coupled to adenylyl cyclase via G s and binding of epinephrine to this receptor leads to increased cyclic AMP synthesis. This is certainly the case in some situations. Binding of a stimulatory G alpha (G s) enhanced activity while binding of an inhibitory G alpha (G i) inhibited cyclase activity. These enzymes differ considerably in regulatory properties and are differentially expressed among tissues, adding support to observations that support a very complex model of interactions that regulate cyclic AMP production.Įarly studies indicated that cyclase activity was regulated primarily by interactions with alpha subunits of heterotrimeric G proteins, which are activated through G protein-coupled receptors. There are at least nine isoforms of adenylyl cyclase, discovered by cloning of full-length cDNAs. When adenylyl cyclase is activated, it catalyses the conversion of ATP to cyclic AMP, which leads to an increase in intracellular levels of cyclic AMP. This form of the enzyme appears to be activated by bicarbonate ion. Two catalytic domains extend as loops into the cytoplasm, as depicted in the figure to the right.Ī soluble (non-membrane bound) form of adenylyl cyclase has recently been characterized in mammalian sperm. Regulation of intracellular concentrations of cyclic AMP is largely a result in controlling adenylyl cyclase.Īdenylyl cyclases are integral membrane proteins that consist of two bundles of six transmembrane segments. Cyclic AMP functions as a second messenger to relay extracellular signals to intracellular effectors, particularly protein kinase A. AMP has been approved by the Food and Drug Administration as a 'Bitter Blocker' additive to foodstuffs.Adenylyl cyclase is the enzyme that synthesizes cyclic adenosine monophosphate or cyclic AMP from adenosine triphosphate (ATP). 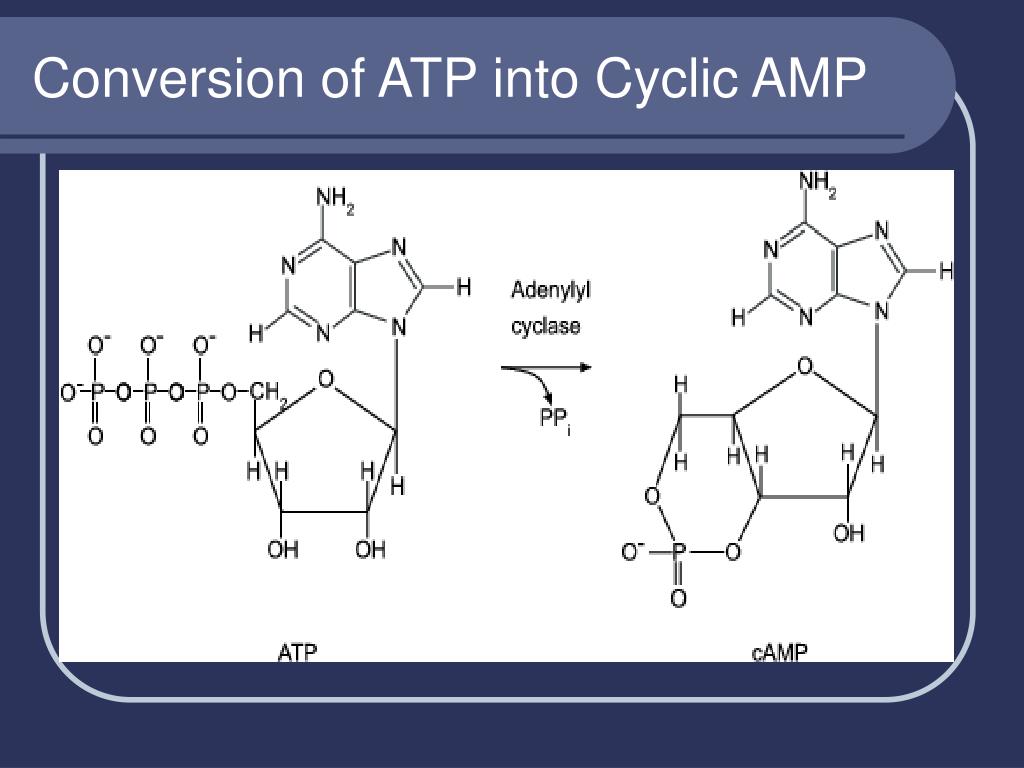
This makes lower-calorie food products more palatable, making AMP potentially a lucrative solution for food manufacturers as they respond to pressure from consumers and regulators concerned about social trends towards obesity. To human tastes, the bitterness-suppressing quality of AMP interprets as food seeming 'sweeter'. cAMP plays an important role in intracellular signaling. Within certain cells the enzyme adenylate cyclase makes cAMP from ATP, and typically this reaction is regulated by hormones such as adrenaline or glucagon. In a catabolic pathway, adenosine monophosphate can be converted to uric acid, which is excreted from the body.ĪMP can also exist as a cyclic structure known as cyclic AMP (or cAMP). When RNA is broken down by living systems, nucleoside monophosphates, including adenosine monophosphate, are formed.ĪMP can be regenerated to ATP as follows:ĪMP + ATP → 2 ADP (adenylate kinase in the opposite direction)Ģ ADP + 2 P i → 2 ATP (this step is most often performed in aerobes by the ATP synthase during oxidative phosphorylation)ĪMP can be converted into IMP by the enzyme myoadenylate deaminase, freeing an ammonia group. Or AMP may be produced by the hydrolysis of one high energy phosphate bond of ADP:ĪMP can also be formed by hydrolysis of ATP into AMP and pyrophosphate: 4 Application as a bitterness suppressorĪMP can be produced during ATP synthesis by the enzyme adenylate kinase by combining two ADP molecules:.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
